[Editor’s Note: This page was originally published on June 28th, 2010 on the ghginstitute.org blog. The post, in its current form, has been edited to include content as of April 1st, 2015.]
What is a global warming potential? And which one do I use?
This question is not as silly as it may seem. This blog post is consistently the most frequently visited page on our website. It is a topic so fundamental to carbon management that many practitioners are probably afraid to seek clarification out of fear of looking uninformed. Since not everyone working on managing greenhouse gas (GHG) emissions has studied atmospheric chemistry (I admit I have, but wouldn’t expect the range of folks working on these issues to have the same background), I am updating our primer on the topic.
But first, you should read my previous blog post on greenhouse gases, which has also been updated.
I’m going to skip over the underlying physics and chemistry because it is not necessary to engage at that level of scientific technicality to be an intelligent user of GWP values. (If you want to dig into the science more, you can refer to the latest IPCC assessment report published in 2013 — see Chapter 8 of the Working Group I report.)
Global Warming Potentials (GWPs) are a quantified measure of the globally averaged relative radiative forcing impacts of particular greenhouse gases. It is defined as the cumulative radiative forcing – both direct and indirect effects – integrated over a period of time from the emission of a unit mass of gas relative to some reference gas (IPCC 1996). Carbon dioxide (CO2) was chosen by the IPCC as this reference gas and its GWP is set equal to one (1). GWP values allow you to compare the impacts of emissions and reductions of different gases.
So to be clear, GWP values are applied to units of mass (e.g., kilograms, pounds, metric tons, etc.), not to units of volume (e.g., cubic meters, cubic feet, liters).
There are three key factors that determine the GWP value of a GHG:
the gases absorption of infrared radiation,
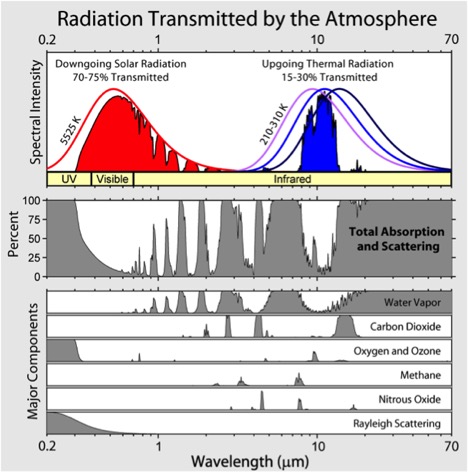
where along the electromagnetic spectrum (i.e., what wavelengths) the gas absorbs radiation, and
the atmospheric lifetime of the gas
We typically only use GWP values for gases that have a long atmospheric lifetime (i.e., in years). Because only these gases last long enough in the atmosphere to mix evenly and spread throughout the atmosphere to form a relatively uniform concentration. GWP values are meant to be “global,” as the name implies. So if a gas is short-lived and does not have a global concentration because it is destroyed too quickly to even mix throughout the atmosphere, then it can’t really have a GWP.
Specifically, the gases with relatively long atmospheric lifetimes that tend to be evenly distributed throughout the atmosphere, and therefore have global average concentrations, are CO2, CH4, N2O, HFCs, PFCs, SF6, and NF3. (There are numerous other more obscure chemicals you can investigate in the IPCC AR5 report chapter). The short-lived gases such as water vapor, carbon monoxide, tropospheric ozone, other ambient air pollutants (e.g., NOx, and NMVOCs), and tropospheric aerosols (e.g., SO2 products and black carbon) vary spatially, and consequently, it is difficult to quantify their radiative forcing impacts.
Some GWP values may account for indirect as well as direct effects. Indirect radiative forcing occurs when chemical transformations involving the original gas produce a gas(es) that is/are also a GHG, or when a gas influences other radiatively important processes such as the atmospheric lifetimes of other gases.
In sum, the higher the GWP value the more infrared radiation the gas will tend to absorb over its lifetime in the atmosphere, leading to more warming. Now, there are three more complications to this story.
The first is that gases will absorb certain wavelengths of radiation. GHGs each absorb in a given “window” of the spectrum. The more that window is filled up, the less there is to absorb. So, as concentrations of certain gases increase they can saturate that wavelength, leaving no more radiation for additional concentrations of gas in the atmosphere to absorb.
The second complication is one that occasionally trips people up. Remember above when we defined GWP by saying “cumulative radiative forcing…integrated over a period of time”? Well, that means that we have to define a time period for the integration to occur. You have to know what the integration period is to make sure you are using the correct GWP. The typical periods that the IPCC has published are 20, 100, and 500 years (the latest report quit publishing values for 500 years).
Now, to be clear, everyone pretty much universally uses 100 year GWP values, so you often never see the time period even cited. It is just assumed you know it is 100 years. But occasionally, someone will use something different, not realizing that they are breaking convention. It is also possible to compute an infinite time horizon GWP value, which would basically mean that accounted for every bit of radiative forcing of every molecule of gas as long as it existed in the atmosphere.
The last complication relates to the fact that the IPCC keeps updating its GWP values with each of its scientific assessment reports. It makes sense to update GWP values as our scientific understanding improves. However, the problem is that people are using and making commitments based on GWP values while these revisions are taking place. So, say a company or a country commits to reducing its emissions by 10% and achieves that goal. Then all of a sudden GWP values change and now they no longer make the goal using the new GWP values (due to the mix of different GHGs they emit and reduce). It would be like moving the net after you already kicked the ball towards the goal.
For this reason, the Kyoto Protocol fixed the use of GWP values published by the IPCC in 1996 in its Second Assessment Report. Since then the IPCC has updated its GWP values three times, in 2001, 2007, and 2013. The result has been a proliferation of GWP values out there that leads to a lot of confusion.
The major causes for the IPCC’s updates to GWP values involved new laboratory or radiative transfer results, improved atmospheric lifetime estimates, and improved calculations of CO2 radiative forcing and CO2 response function. When the radiative forcing of CO2 is updated, then the GWPs of the other gases relative to CO2 also change. The IPCC has also added numerous new, and rarely used, gases to its list of GWPs.
The result of the varying time periods and the regular updates by the IPCC is a complicated state of affairs. This table presents GWP values for the most common GHGs (there are many more if we listed all the HFCs, PFCs, halogenated alcohols and ethers, and other trace gases). As you can see in this table, each gas has a number of GWP values that you could choose.
But the truth is, contrary to what a layperson might expect, we typically only use values over a 100 year time period, even though some gases have lifetimes of thousands of years.
Until recently, we also almost always used the values published by the IPCC because they were adopted by the UNFCCC and Kyoto Protocol. However, now with the Kyoto Protocol effectively over, the UNFCCC has now adopted the IPCC 2007 AR4 values for international reporting. Yet, we now have newer values from the IPCC 2013 AR5 report.
So, after many years of stability based on the 1996 IPCC values, we have entered a period of confusion surrounding what vintage of GWP values should be universally applied so all climate change programs and policies around the world are consistent in their emissions accounting.
I’ve highlighted in red the values currently adopted by the UNFCCC national emissions reporting. And highlighted in green are the most recent values from the IPCC AR5. I wish I could tell you which one to use. As a default, I would recommend you use the red 2007 values to be consistent with the UNFCCC. But recognize that an agreement later this year in Paris could adopt the new 2013 AR5 values.
And if you are still using the old 1996 SAR values, it is probably time to update.
Table: Global Warming Potential Values from the IPCC for some key GHGs
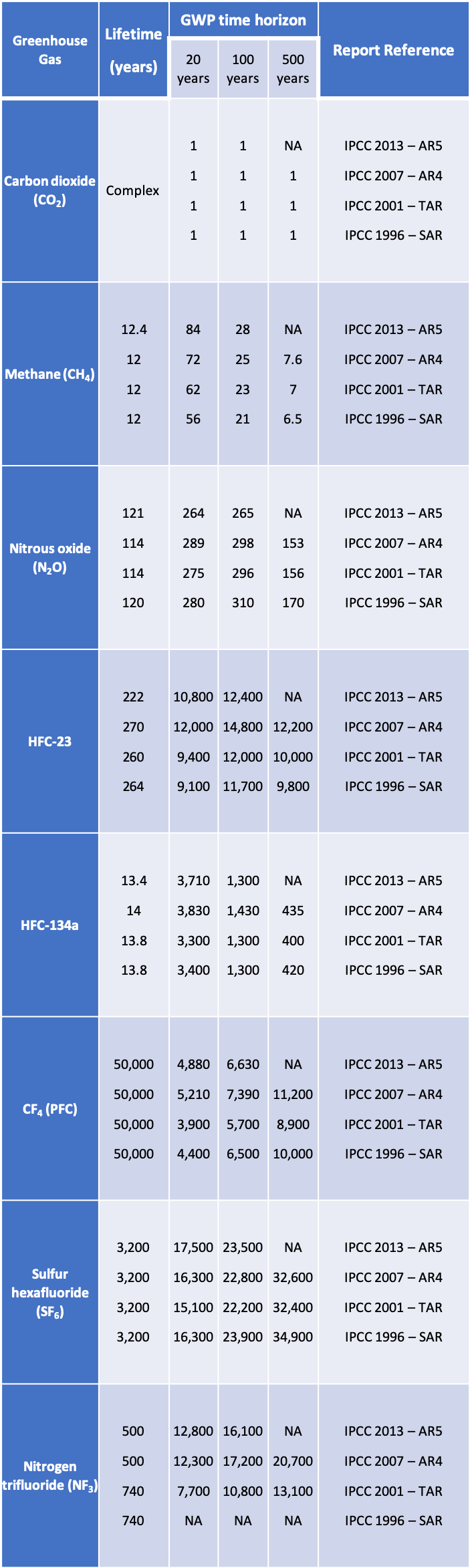
NA: Not available
Row 1: 2013 IPCC AR5(See Chapter 8 of Working Group I report)
Row 2: 2007 IPCC AR4 (See Chapter 2 of Working Group I report)
Row 4: 2001 IPCC TAR (See Chapter 6 of Working Group I report)
Row 4: 1996 IPCC SAR (See Chapter 2 of the Working Group I report)
To wrap things up for the sake of being thorough, the relationship between the mass of gas and the mass of CO2 equivalent can be expressed as follows:
mass CO2 Eq. = (mass of gas) x (GWP)
Where:
mass CO2 Eq. = mass (e.g., metric tons) of Carbon Dioxide Equivalents
GWP = Global Warming Potential
So the calculation is easy. Just multiply the mass of your gas by its GWP value to get CO2 equivalent emissions. Be sure to label the resulting emissions not as CO2, but as “CO2-equivalents.” Note, that this is not carbon, but CO2. The ratio of carbon to CO2 is 12/44. So, if you hear someone talking about carbon emissions make sure you have them clarify what they are actually talking about.
And in case you were wondering, according to the IPCC, GWPs typically have an uncertainty of roughly ±35 percent, though some GWPs have larger uncertainty than others.
Lastly, there is one more confusing issue, which I will only touch on briefly. There are numerous gases like chlorofluorocarbons (CFCs), hydrobromocarbons (e.g., methyl bromide), and halons that deplete stratospheric ozone. These gases are being phased out under the Montreal Protocol and related international agreements. They are also GHGs, although their impact on radiative forcing is even more complex because stratospheric ozone is also a GHG. So, ozone-depleting substances (ODSs) have both positive and negative radiative forcing effects. We generally do not include them in GHG emission inventories because they are being phased out, although some carbon offset projects are crediting the destruction of ODSs.
